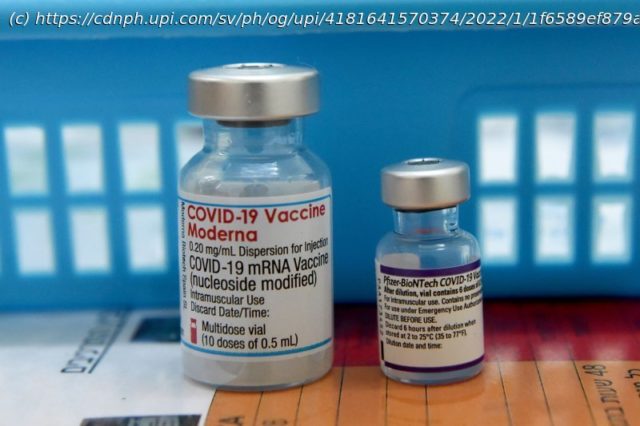
The U.S. Food and Drug Administration amended the emergency use authorization for the Moderna COVID-19 vaccine to shorten the interval between primary vaccination and boosters to five months.
Timeline words data
Options
Restore all
Clear all